I first heard about the mandate for QR code on drugs three years ago.
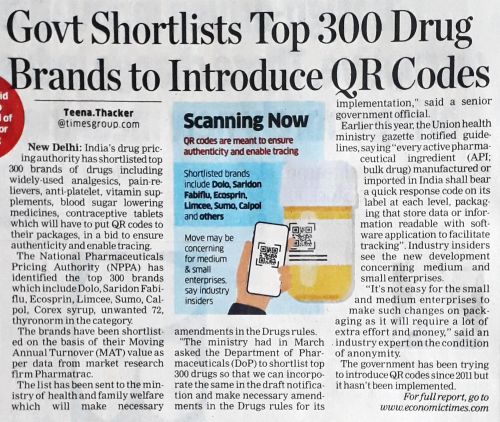
According to this article in Economic Times dated 3 January 2022, National Pharmaceuticals Pricing Authority (NPPA) shortlisted Top 300 drugs for insertion of QR codes on their packages. These drugs included analgesics, pain relievers, anti-platelets, vitamin supplements, diabetes and contraceptives. The goal of the regulation was to ensure authenticity and enable tracing of drugs. Apparently, the government has been trying to introduce QR codes since 2011.
While I’ve not heard of any name for this regulation, I’ll call it Reg DrugsQR for easy referenceability.
I first noticed a QR code on CELIN 500, a vitamin supplement, in November 2022.
I scanned the QR code, hoping to see authenticating information. However, all I saw was a PDF with a remark that it was meant “For the use of a Registered Medical Practitiioner, Hospital and Laboratory only“. I muttered duh to myself and moved on.
I didn’t see anything in the media on this topic for the next two years. Then, on 5 October 2024, Economic Times carried an article about QR code on cancer medicines.
Going by the headlines and the first half of the body of the article, this seemed to be a new regulation mandating QR codes only for cancer drug strips and vials. However the second half of the article segued into top 300 drugs including analgesics, etc. To add to the confusion, the article invoked the name of a different regulator viz. Drugs Technical Advisory Board (DTAB).
I was not alone in being confused. According to the last paragraph of the article:
Pharma companies and lobby groups had expressed concerns over multiple sets of directives regarding tracing and tracking being issued by various departments and had asked for a single QR code system.
I posted the following tweet soon after:
@s_ketharaman: Reg QR Code on Drugs has been talked about for 10+ years but has still not been implemented apparently due to stonewalling by pharma industry.
Once again, there was silence on this topic for another year.
Then, on 21 October 2025, Pune Mirror reported that the central government made QR codes mandatory for about 300 drugs.

There seemed to be a sense of finality in the tone of this article. According to it:
- Government of India via Drugs Controller General of India (DCGI) has made QR codes mandatory on the packaging of top 300 drugs in India with effect from 1 August 2025.
- The list includes common medicines like Allegra, Calpol, Dolo, Shelcal, and Metfal.
- The objective of the mandate is to curb production and sale of fake medicines, estimated to be 20% of all drugs sold in the country.
- Citizens will be able to verify the genuineness of a medicine directly through their smartphones. This would help build greater trust among patients, doctors and pharamacists.
- By scanning the QR code, consumers will instantly access all official information about the medicine on their smartphones such as Unit Product Identification Code, Brand Name, Generic Name, Manufacturer’s Address, Batch Number, Manufacturing Date, Expiry Date, and License Number.
- Since the article does not make any mention of cancer-only drugs, I’m guessing the pharma industry’s plea to have a single QR code system has been answered by the government.
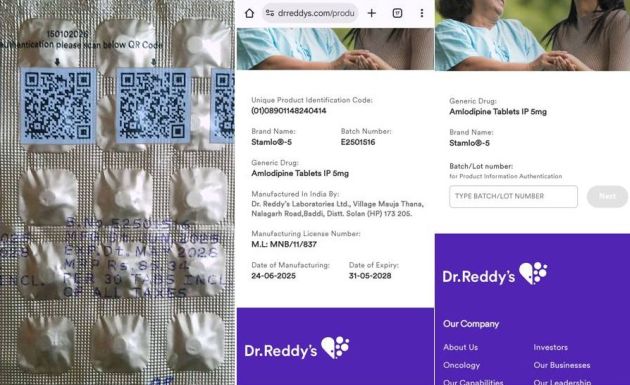
Soon afterwards, I noticed QR codes on STAMLO (see footnote 1).
When I scanned the QR code, I was taken to a landing page shown in the following exhibit.
As you can see, the landing page provides all the details like Unit Product Identification Code, Brand Name, Generic Name, etc. as stipulated by Reg Drugs QR. Accordingly, this drug complies with the letter of Reg DrugsQR (Not legal advice).
Next, let’s see how the QR code achieves the goal of enabling consumers to verify the genuineness of a medicine directly through their smartphones.
As long as the consumer gets a prescription from the doctor with the name of the medicine, name of manufacturer, URL of manufacturer’s website, s/he can scan the QR code on the drug’s packaging, look up the landing page and authenticate the drug (see footnote 2).
| PRESCRIPTION | QR CODE LANDING PAGE | CHECK | |
|---|---|---|---|
| Name of Medicine | STAMLO | STAMLO | YES! |
| Name of Manufacturer | DR. REDDY’S | DR. REDDY’S | YES! |
| URL of Manufacturer’s Website | drreddys.com | drreddys.com | YES! |
The consumer can do two more things to gain confidence about their purchase:
- Look at the Expiry Date on the landing page and verify that it’s in the future, thus indicating that s/he has not been sold an expired drug.
- Compare the Manufacturing Date on the landing page with the date of purchase. As long as the former is earlier than the latter, this provides an additional level of comfort that the drug is genuine (see footnote 3).
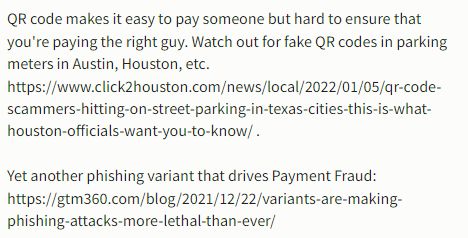 The above of course assumes that the fakester does not manage to print his fake QR on the packaging (see footnote 4).
The above of course assumes that the fakester does not manage to print his fake QR on the packaging (see footnote 4).
Given the modus operandi followed by fakesters of cancer drugs described in the above mentioned article – “refill empty vials of expensive anti-cancer medicine with counterfeit drugs” – this is a nontrivial assumption but one that’s safe to make for all but the very expensive drugs. For cheap drugs like Allegra, Calpol, Dolo, Shelcal, and Metfal, Stamlo, etc., I doubt if the returns would be worth the trouble and risk for the fakesters.
Interestingly, the CELIN 500 strip I bought last month did not have a QR code. As I mentioned earlier, this was the first medicine on which I’d seen a QR code three years ago. I’m guessing vitamin supplements has been dropped from the original list of 300 drugs covered under Reg DrugsQR.
FOOTNOTE(S):
- QR codes, as in plural. However, they all went to the same landing page.
- Not sure how a prescription that contains all these details of a specific medicine will comply with another law that stipulated that doctors should only prescribe for unbranded generic drugs. Maybe that law has still not come into effect?
- I’ve seen complaints from consumers on X fka Twitter that they have received goods ordered on Zepto and other quick commerce companies that bear a manufacturing date that’s after the date of purchase, which is a telltale sign of something fishy.
- According to this YouTube Shorts video, fakesters have replaced some merchant’s UPI QR code with their own!